Discover FMEA in less than four minutes!

The FMEA web application that truly structures and compiles your FMEA studies!
Robust Engineering Software and TEEXMA for Quality module
Design, Means, and Process FMEA studies; PRA; system risks views; control plans; flow charts; workstation charts; and key characteristics.
A standard, customizable FMEA tool
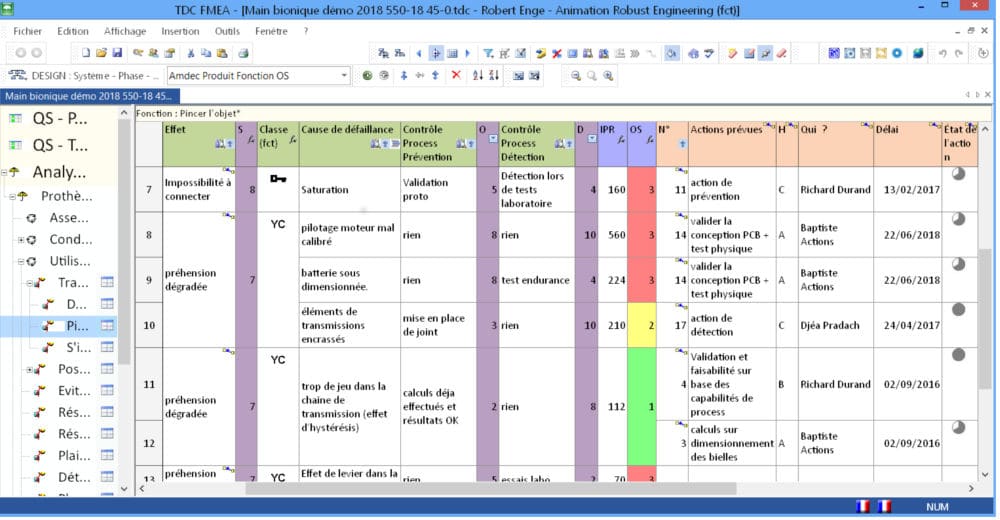
Robust Engineering Software’s FMEA tool manages different FMEA risk studies under the same software licence: Design FMEAs, Process FMEAs, DRBFMs, flow charts/manufacturing flow charts, control plans, action plans, workstation charts, and special characteristics.
You can choose to work with only one type of FMEA (Design FMEA, for example) or to run an integrated and coherent Design/Process FMEA series within the same study using propagations and control of the special characteristics, the corresponding scoring, and so on.
The tool proposes standardized reference documents:
- The classic AIAG 4th Edition reference manual (2009), the VDA Volume 4 FMEA Manual,
- or the new 2019 AIAG & VDA Handbook – see the AIAG-VDA software or our webinars.
This FMEA tool can be totally customized to your work habits and methods—at the key-in interface level (charts, thresholds, scoring, etc.) as much as at the FMEA documents and indicators level.
Knowllence’s FMEA tool comes in a variety of languages. With multi-language management, you can designate each of your datum and your libraries in as many languages as needed – including Chinese, Polish, and Czech, and many more.
Coherence and sharing of all your FMEA data
The coherence of data and documents is ensured within a single database through knowledge sharing between the various tools throughout the design process—from the product to the process:
- Within the FMEA tool: between the Design FMEA, DRBFM, Process FMEA, control plan, flow chart, and special characteristics.
- With additional design modules for functional analyses in Robust Engineering Software in order to carry out a methodologically “correct” and complete FMEA study, the FMEA tool shares pertinent information about need.
- The process occurrence statistics and the links to the causes of malfunction can be retrieved from your non-compliance management or production monitoring (SPC) products to enhance the Process FMEA studies (dynamic connector).
Save time and return on investment with your FMEAs
Our customers have told us that they have earned a quick return on investment with the FMEA tool, as the time spent creating their FMEA studies has been cut up to 50% within the first ten months after software training.
The FMEA tool also provides a common work platform that makes communication between the various project teams easier: R&D, Quality, Design Office, Industry, Production, Maintenance, and even the Marketing Department if the product risks identified have an impact on your business.
By using the FMEA tool and method, you’ll see an improvement in accuracy in your projects that will motivate everyone involved. It will become a veritable productivity tool that will boost your image in the eyes of your very demanding customers.
The FMEA tool facilitates the operational direction of action plans using indicators, Dynamic HTML documents, and informational and reminder emails.
You can create structured, standardized, and uniform FMEA studies for the project teams and your whole company—which, in turn, boosts efficiency, simplifies reuse, and provides coherent deliverables to your customers.
The tree elements are displayed using modifiable colour codes according to the criticality thresholds. This helps you target the key points to work on instantly that are in line with Lean methods.
Advantages: Libraries and generic FMEAs
Knowllence’s FMEA tool can create cumulative generic FMEAs that can be enhanced as projects move along. Consequently, any changes can be easily propagated across the other projects in progress.
The goal is to free yourselves from an overabundance of study names or past projects by organizing all cumulated experience into a single database. Once the functions, components, and process operations have been chosen, you will be able to see the origin of each piece of information that has been touched (date, author).
This helps you effortlessly create references and manage your know-how within your company—a long-term and reliable effort through the traceability of each datum individually. With a simple right-click of your mouse, you’ll see the origin of a datum and decide whether to integrate it or not into your project.
- You can create as many effect, mode, cause, and means of prevention libraries, etc., as you need. These libraries are linked together and let you choose from the usual failure modes linked to a function/component type, as well as the usual causes associated with a failure mode, and so on. All of your studies will have the same technical lexicon, and your accuracy will increase through the tool’s prompts—in any of the languages you work with.
- Of course, you can add to and enhance these multi-lingual libraries if you have the user rights.
- You can create complete studies from generic Design or Process FMEAs, with all of the links that have already been established between the two. You can also integrate generic elements chart by chart to create a generic study.
- After formalizing changes or additions (incidents, 8D, etc.) in a generic study, you can then choose to extend them to other products, which guarantees they are systematically taken into account.
You’ll be able to manage your company references in a dynamic, continual manner—under the watchful eye of a responsible party who validates each before it is transmitted. This helps contribute to your knowledge management strategy.
Directing action plans
The FMEA tool contributes to setting up a paperless, digitally driven factory:
- The program provides easy monitoring of actions through a system with a one-click filter, correspondence, indicators, and informational and reminder email alerts.
- The system to manage actions is centralized either in the program, our cross-functional tool Action Facilitator, or your company’s action plan management system (dynamic connector).
- When actions are tagged following an incident or proven problem in the generic study, the FMEA tool duplicates them to all of the similar product references implicated, so it’s the tool that does the work.
- The list of actions is available in Excel, PDF, or HTML formats. They are sorted by the responsible party, status of the actions, or deadline. The HTML format allows filtering and sorting of the status of the actions, responsible parties, deadlines, etc.
- HTML documents benefit from all the advantages found with browsers such as Chrome, including automatic translation functions.
Managing control plans
Since they are managed within the same database and share the same data, control plans are managed in coherence with Process FMEAs. You can also choose to create the controls and the characteristics using detection within the Process FMEA,
which limits differences between the Process FMEA and the control plan, and therefore reduces the risk of non-compliance during audits.
The control plans can be managed at the prototype, pre-production, or production level.
Find out more about our FMEA tool control plans
Monitoring special characteristics
By using a centralized database, the FMEA tool monitors all of the critical characteristics, including the functional criteria, component characteristics, and process operation parameters. Coherence is guaranteed as automatic alerts are sent out if changes happen on any level. The FMEA tool also ensures coherence and propagation of the scoring of effects associated with special or critical characteristics.
Learn more about management of special characteristics using the FMEA tool
Traceability within the FMEA tool
The FMEA program manages traceability (otherwise known as the log history) at the level of each datum. You can then simply filter the results to find any information linked to a person or a date at any time. You’ll get an immediate readout on what has happened since the last meeting or previous audit, for instance.
You can:
- Find out who modified contents (wording) and when through a simple right-click on each cell
- Access the entire log history of actions carried out on the study
As a new and unparalleled role found on today’s market, storyboards take this tracing information and use them to go above and beyond indicators and reports through a dynamic approach. It’s like making a full-length film out of a series of photos with the aim to create a visual aid for self-auditing and improve processes and best practices. For example, you will be able to visualize the progression of the changes to requirements over time (a given date, a step/milestone in the development of a product) to identify the impacts of these changes better.
A few of the FMEA tool features
- Customization of guide rules (alerts, severity and effect links, etc.) and ease of entering information (vocabulary, columns, contextual help, etc.)
- Customization of communication materials: indicators, OS/DS criticality, summary reports, action plans as Gantt charts, self-inspection procedures, workstation charts, elements for the supporting design file, etc.
- Graphic filters and sorting tools, including in HTML-based communication materials: all changes that have happened in the study from or on a specific date, all data specific to a project, etc.
- Graphs for all the digital data, radar charts
- Advanced cut/paste functions (tree branches and their corresponding charts), including from one study to another
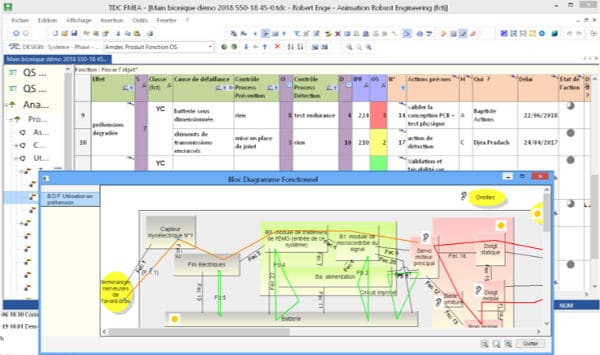
- Cross-category charts: Several charts linked to each other at all the different levels of the tree. For example, to detail the OS and OS/DS scoring or to link the Process FMEA/control plan/workstation charts
- Searching for causes through Ishikawa diagrams
- Going from CAD to FMEAs: retrieve dimensions and tolerances in your CAD designs when beginning FMEAs with Infra CONVERT
- Drag and drop directly from Excel to retrieve your existing studies
- Manage several languages for the data in the study. French, English, German, Spanish, and Portuguese. Key in is possible in Chinese, Polish, etc.
- Import/Export with Word, Excel, Access, or XML
- Immediate access to the view of the functional block or SADT diagrams when creating Design FMEAs if the Structure functional block diagram tool is activated.
- Customizable management of rights and roles so that each person only has access to the information they need and to restrict the possibilities for changes if needed.
- Can be interfaced with other programs: quote for connectors – for study data and libraries.
Knowllence’s FMEA software can be fully customized to your work habits or to your needs for consolidating information from different sites.
Each person has the point of view that concerns him or her: designer, FMEA animator, action pilot, simple work group member, manager, etc.
Do not hesitate to contact us!